X-ray photons are produced when electrons in the outermost shells of an atom drop to the inner shells. Like all electromagnetic radiation, X-rays are made of photons. The study of atomic energy transitions enables us to understand X-rays and X-ray technology. Proteins near the surface of the skin emit a characteristic blue light.įigure 8.20 A scorpion glows blue under a UV lamp. Figure 8.20 shows a scorpion illuminated by a UV lamp. 
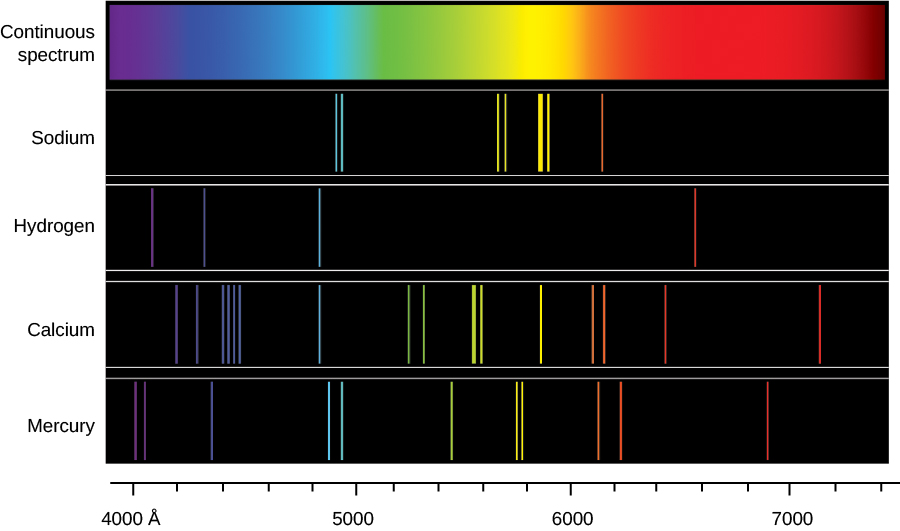
Fluorescent lights are more efficient in converting electrical energy into visible light than incandescent filaments (about four times as efficient). Fluorescent dye in clothes can make colors seem brighter in sunlight by converting UV radiation into visible light. Some of these photons may be in the visible range. The electron can drop back to the ground state, emitting a photon of the same energy that excited it, or it can drop in a series of smaller steps, emitting several low-energy photons. Once excited, the electron “de-excites” in two ways. Atomic Fluorescenceįluorescence occurs when an electron in an atom is excited several steps above the ground state by the absorption of a high-energy ultraviolet (UV) photon. However, a sensitive spectrometer can measure the difference. The energy difference Δ E Δ E is about 0.1% (1 part in 1000) of this average energy. Interestingly, mercury has two separate energy-level diagrams these diagrams correspond to two net spin states of its 6 s (valence) electrons.Į = h c λ = ( 4.14 × 10 −15 eVs ) ( 3.00 × 10 8 m/s ) 589.30 × 10 −9 m = 2.11 eV. The valence electron of sodium moves in the electric field of a nucleus shielded by electrons in the inner shells, so it does not experience a simple 1/ r Coulomb potential and its total energy depends on both n and l. The energy-level diagram for hydrogen is similar to sodium, because both atoms have one electron in the outer shell. (These levels are not vertically displaced, because the energy splitting is too small to show up in this diagram.) Likewise, spin-orbit coupling splits the n = 3 n = 3 states into three angular momentum states ( s, p, and d). However, spin-orbit coupling splits the n = 2 n = 2 states into two angular momentum states ( s and p) of slightly different energies. If we neglect electron spin, all states with the same value of n have the same total energy. The hydrogen atom has the simplest energy-level diagram. For comparison, hydrogen energy levels are shown in the sodium diagram. Figure 8.19 Energy-level diagrams for (a) hydrogen, (b) sodium, and (c) mercury. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
